Evaluation of normalized T1 signal intensity obtained using an automated segmentation model in lower leg MRI as a potential imaging biomarker in Charcot– Marie–Tooth disease type 1A
Deep Learning Medical ImagingPosted by mhb on 2025-11-19 11:21:51 |
Share: Facebook | Twitter | Whatsapp | Linkedin Visits: 175
Introduction
Charcot–Marie–Tooth disease type 1A (CMT1A) is the most common hereditary neuromuscular disorder and is characterized by progressive weakness, distal muscle wasting, and sensory loss. Because the disease progresses slowly, there is a growing need for objective biomarkers that can quantify severity and track progression. T1-weighted MRI has long been used to evaluate muscle denervation and fat replacement, but direct signal intensity values are highly dependent on acquisition parameters, limiting their usefulness for quantitative analysis. Signal intensity normalization can overcome this limitation by adjusting MRI values to a consistent reference.
Deep learning–based automated muscle segmentation now allows efficient and reproducible extraction of muscle-specific MRI measurements, eliminating the requirement for labor-intensive manual segmentation. In this study, normalized T1 signal intensity was calculated for the lower-leg muscle compartments of patients with CMT1A using an automated segmentation model. The goal was to examine whether these normalized measurements could serve as quantitative imaging biomarkers of intramuscular fat replacement by evaluating their correlations with clinical severity measures.
Methods
Patient Population
This retrospective study included 107 genetically confirmed CMT1A patients who underwent lower-leg MRI between 2015 and 2023. Patients with missing clinical data, poor MRI quality, or prior lower-extremity surgery were excluded.
MRI Acquisition
All MRIs were obtained using a 3T system with axial T1-weighted turbo spin-echo sequences of the lower legs. Imaging parameters included 612 ms repetition time, 11 ms echo time, 3 mm slice thickness, and a 284 × 379 mm field of view.
Automated Segmentation and Signal Extraction
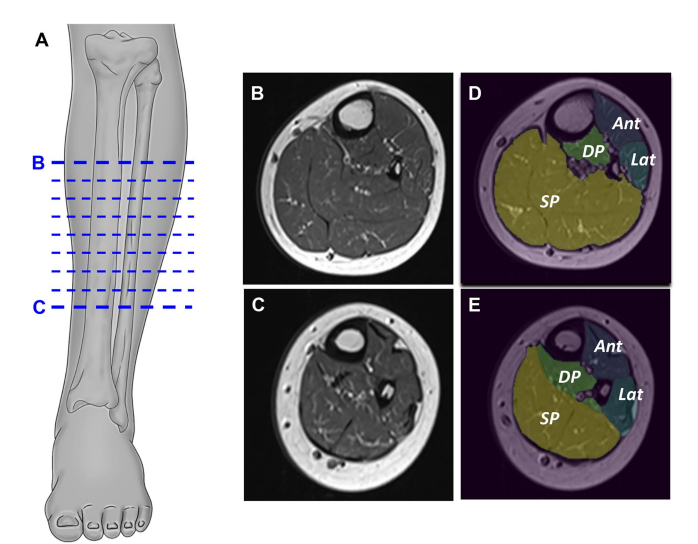
A deep learning U-Net–based segmentation model previously trained on manually labeled datasets was used to segment four lower-leg muscle compartments: anterior, lateral, deep posterior, and superficial posterior. For each slice:
-
Mean signal intensity of each muscle compartment was calculated.
-
A reference signal intensity was computed from fat-rich structures such as subcutaneous fat and bone marrow while excluding muscle and background.
-
The compartment signal was divided by the reference signal to compute normalized intensity.
-
Slice-level values were averaged to produce a single mean normalized signal intensity (MNSI) per compartment.
Higher MNSI values indicate greater fat replacement. Total MNSI was also calculated by combining all muscle compartments.
Clinical Measures
Clinical severity was evaluated using Charcot–Marie–Tooth Neuropathy Score version 2 (CMTNSv2), functional disability scale (FDS), 10-meter walk test (10-MWT), and 9-hole peg test (9-HPT).
Statistical Analysis
Partial correlation analysis was performed to examine associations between MNSIs and clinical measures, adjusting for age and body mass index because both influence intramuscular fat content.
Results
The study included 56 men and 51 women with a mean age of 41 years. Clinical severity varied widely across the cohort. MNSIs differed among muscle compartments, with the lateral compartment showing the highest fat-related intensity and the deep posterior compartment the lowest.
Significant positive correlations were observed between MNSI values and all clinical severity measures:
-
CMTNSv2
-
FDS score
-
10-MWT time
-
9-HPT time
All p-values were < 0.05 after adjustment for age and BMI. The strongest correlation was between the anterior compartment MNSI and FDS score (r = 0.57). These results indicate that higher normalized T1 signal intensities correspond to more severe clinical impairment.
Conclusion
Normalized T1 signal intensities derived from automated muscle segmentation on standard MRI show strong correlations with established clinical severity measures in CMT1A. This approach provides a simple and widely applicable method for quantifying intramuscular fat replacement without requiring specialized MRI sequences. The findings support the use of normalized T1 intensity as a potential imaging biomarker for disease severity in CMT1A. Future studies comparing normalized T1 measures with Dixon-based fat-fraction imaging and examining longitudinal changes would help confirm its value as a tool for monitoring disease progression.
Search
Categories
Recent News
- Clinical application research on the titanium metal metatarsal prosthesis designed through FEA and manufactured by 3D printing
- Manually weighted taxonomy classifiers improve species-specific rumen microbiome analysis compared to unweighted or average weighted taxonomy classifiers
- A deep learning based smartphone application for early detection of nasopharyngeal carcinoma using endoscopic images
- sCIN: a contrastive learning framework for single-cell multi-omics data integration
- CustOmics: A versatile deep-learning based strategy for multi-omics integration
- Tracking temporal progression of benign bone tumors through X-ray based detection and segmentation
- mCNN-GenEfflux: enhanced predicting Efflux protein and their super families by using generative proteins combined with multiple windows convolution neural networks
- Evaluation of normalized T1 signal intensity obtained using an automated segmentation model in lower leg MRI as a potential imaging biomarker in Charcot– Marie–Tooth disease type 1A
Popular Paper
- sCIN: a contrastive learning framework for single-cell multi-omics data integration
- CustOmics: A versatile deep-learning based strategy for multi-omics integration
- A deep learning based smartphone application for early detection of nasopharyngeal carcinoma using endoscopic images
- Tracking temporal progression of benign bone tumors through X-ray based detection and segmentation
- Clinical application research on the titanium metal metatarsal prosthesis designed through FEA and manufactured by 3D printing